
Now we obtain two important equations from above derived expressions, namely:īoth equations are known as Gibbs Equation. On the other hand, changes in entropy can be quite straight- forward to calculate. We can rewrite the equation of entropy by using the 1.Law of thermodynamics: We write down here the definition of entropy:Īccording to the 1.Law of thermodynamics for reversible process which is expressed as: 1 mol of He (g) at 10 K and 1 atm pressure or 1 mol of He (g) at 250☌ and 0.2 atm. Predict which substance in each pair has the higher entropy and justify your answer. So if we want to calculate the entropy change ΔS, we can arbitrarily choose some imaginary reversible processes from initial state to final state. Based on the greater freedom of motion available to atoms in a liquid, we predict that the liquid sample will have the higher entropy. Recall that entropy, which is defined as δq rev/T, is a property of state. Download the script: Entropy change of ideal gases Textbook content produced by OpenStax is licensed under a Creative Commons Attribution License. Use the information below to generate a citation. 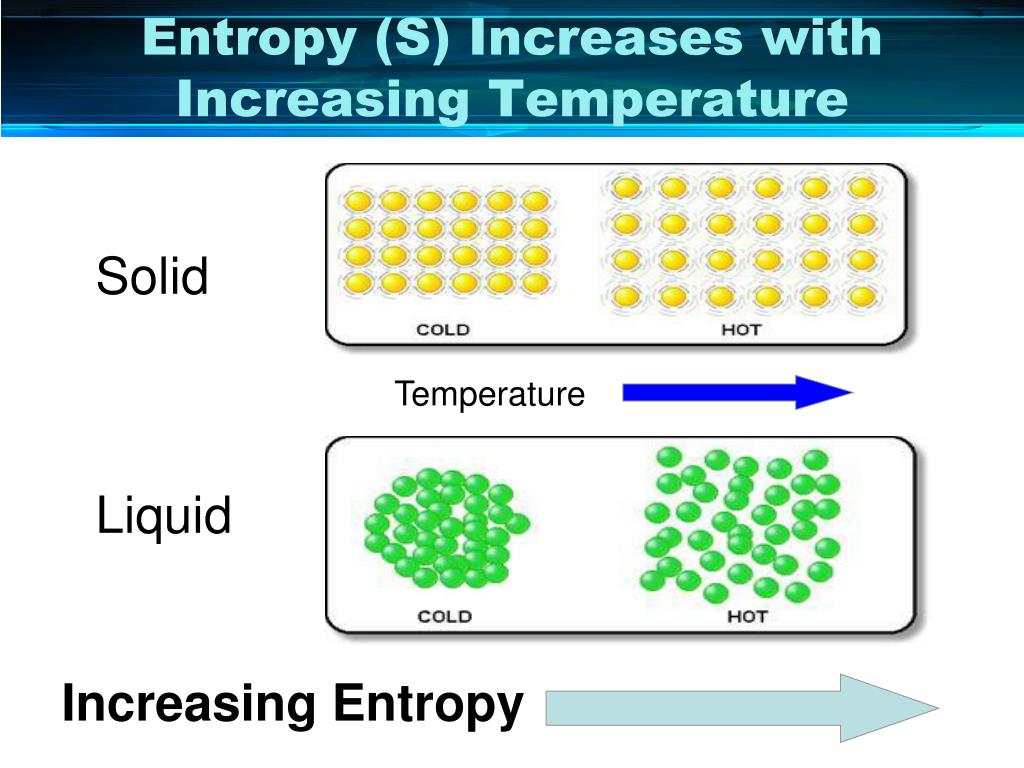
Then you must include on every digital page view the following attribution: But, for an irreversible cycle, world entropy change (net entropy. Because both of system and environment return to their initial states when cycle is completed. 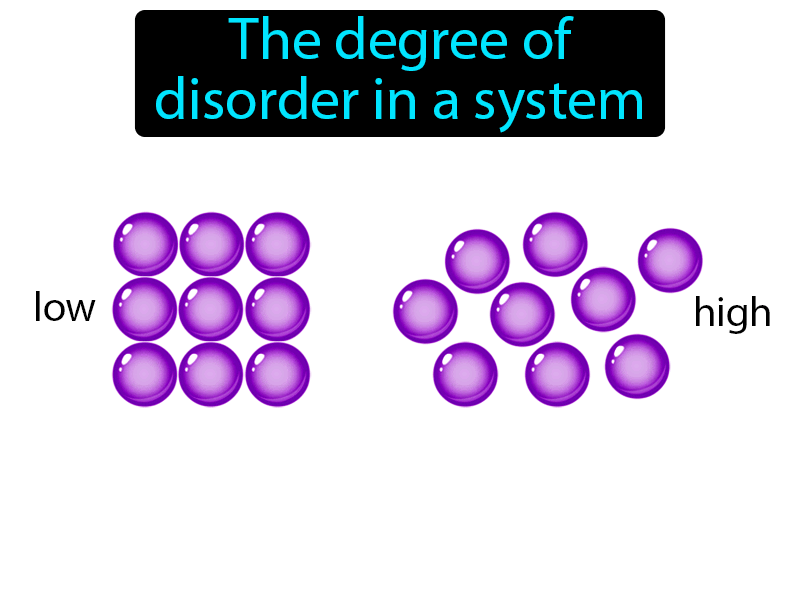
All spontaneous change occurs with an increase in entropy of the universe. The universe tends toward increased entropy. For a reversible cycle, world entropy change is zero. Entropy, S, is a state function and is a measure of disorder or randomness.A positive (+) entropy change means an increase in disorder. In a chemical reaction, the change in entropy can also be attributed to the rearrangement of atoms or ions from one pattern to another. Net entropy change means entropy change of the world (world means system plus environment). Thus, the greater the disorderliness in an isolated system, the higher is the entropy. The change in entropy is positive, because heat transfers energy into the ice to cause the phase change. because of the precise definition of constant pressure heat capacity in thermodynamics: Cp (H T)P C p ( H T) P. Determine the change in the moles of gas for each of the reactions and decide if the entropy increases, decreases, or has little or no change. So, in this case, dqrev dH CpdT d q r e v d H C p d T, where dT d T is the change in the system temperature over the present increment of heat exchanged. 
If you are redistributing all or part of this book in a digital format, Hence, change in entropy does not differ with the nature of the processes either reversible or irreversible. The change in entropy, Srxn, is related to the change in the number of moles of gas molecules, ngas. Then you must include on every physical page the following attribution: If you are redistributing all or part of this book in a print format, That will be the change in entropy for any process going from state 1 to state 2. We just need to find or imagine a reversible process that takes us from state 1 to state 2 and calculate S S for that process. Want to cite, share, or modify this book? This book uses the Thus the change in entropy S S of a system between state 1 and state 2 is the same no matter how the change occurs. The net change in entropy of the system for the transition is The change in entropy for each step is Δ S i = Q i / T i. This can be accomplished experimentally by placing the system in thermal contact with a large number of heat reservoirs of varying temperatures T i T i, as illustrated in Figure 4.15. During each step of the transition, the system exchanges heat Δ Q i Δ Q i reversibly at a temperature T i. The temperatures associated with these states are T A T A and T B, T B, respectively. This modification in the entropy formula gives an indication of a process’s or a chemical reaction’s spontaneity. Imagine a system making a transition from state A to B in small, discrete steps. The following change in the entropy equation can be used to signify the formula for Entropy Changes in the Universe: Suniverse Ssystem + Senvironment. The change in entropy of a system for an arbitrary, reversible transition for which the temperature is not necessarily constant is defined by modifying Δ S = Q / T Δ S = Q / T. DSsyst, the entropy change of the system, represents the change in order of the molecules of the system, similar to what was discussed in Entropy 2. The same equation could also be used if we changed from a liquid to a gas phase, since the temperature does not change during that process either. Δ S = 16.8 kJ 273 K = 61.5 J/K Δ S = 16.8 kJ 273 K = 61.5 J/Kĭuring a phase change, the temperature is constant, allowing us to use Equation 4.8 to solve this problem.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
